
Читайте также:
|
Transcatheter Aortic Valve Replacement Procedure
In transcatheter aortic valve implantation, the patient is “put to sleep” with general anesthesia. In traditional aortic valve replacement, the patient is placed on a heart-lung machine, also known as cardiopulmonary bypass. The chest wall is then opened, and the patient’s heart can be directly accessed to remove the old valve and implant a new valve. With TAVI, a balloon is used to expand the patient’s own aortic valve to make room for the transcatheter valve. The new transcatheter heart valve is then inserted across the patient’s own aortic valve, and another balloon is used to expand the valve in place. There are two main methods used to place the valve, the transfemoral and transapical approach. The decision to use a particular approach is complex, and best discussed with a physician.
Transfemoral Approach
In the transfemoral approach, an incision is placed in the femoral artery in the groin area. A wire is then advanced upwards towards the heart and across the aortic valve. The transcatheter SAPIEN valve is then expanded in place, as shown below.
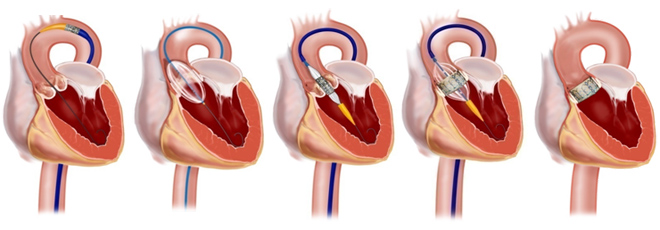
Transapical Approach
In the transapical approach, a small incision is made between the ribs in the chest wall. The valve is then directly inserted across the apex of the heart and into position across the native valve. The valve is then deployed in place, again using a balloon. The incision is then closed with sutures.
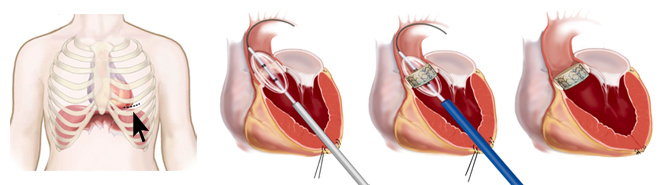
Percutaneous Balloon Aortic Valvuloplasty
Percutaneous balloon aortic valvuloplasty was developed after the successes achieved with mitral and pulmonary valvuloplasties in the 1980s. This procedure provides only modest hemodynamic improvement and is associated with a high incidence of restenosis. Long-term survival after this procedure is not significantly different from the natural history of aortic stenosis.
At present, percutaneous balloon aortic valvuloplasty is done primarily in inoperable patients for the purpose of achieving temporary improvement in quality of life or buying time before more definitive treatment can be performed. The current focus of scientific interest has shifted from balloon aortic valvuloplasty to percutaneous aortic valve replacement.
Percutaneous Aortic Valve Replacement
General considerations
The current design requirements for a permanent catheter-based aortic valve system are formidable and have evolved over the past few years since the first successful percutaneous aortic valve implant by Cribier et al.[2] Design engineers currently favor the use of biologic trileaflet tissue valves, and pericardium has been the valve material of choice.
Percutaneous aortic valve replacement is indicated for high-risk patients with a predicted EuroSCORE greater than 30% and an actual 30-day mortality lower than 10%. It is contraindicated in patients with severe calcification of the aortic valve and severe peripheral vascular disease.
Procedure
The trileaflet valve is sewn or affixed to a stent (or cage) and mounted on a catheter system for deployment. The earliest percutaneous aortic stent valves were large (24 to 26 French), stiff devices, but successive iterations have reduced the system profiles of some devices to 18 French.
The stent valve is deployed either by expansion of the balloon on a balloon-expandable stent or by withdrawal of a sheath that releases a self-expanding stent. The deployed stent valve must remain securely implanted with displacement of the diseased native valve so that the new tissue valve can begin to function immediately.
The hemodynamic performance of catheter-based aortic valves should be similar to that of their surgical counterparts, with minimal transvalvular gradients (< 10 mm Hg) and measured aortic valve areas of 1.5-2.0 cm2. No component of the stent valve can obstruct the native coronary arteries or interfere with the function of the mitral valve apparatus. In addition, the stent valve must be circumferentially apposed to the aortic annulus to prevent paravalvular aortic regurgitation.
Percutaneous Tricuspid Valve Repair
Percutaneous tricuspid valve devices remain in the preclinical stages of development. Tricuspid annular devices must be correctly positioned to avoid obstruction of the coronary sinus ostium or damage to the atrioventricular node.
An advantage of these devices is that they can be deployed under significantly tighter time constraints than other valve devices because right chamber pressures are lower and rapid pacing will not be required. Their disadvantages include the greater fragility of tricuspid valve leaflets, the lack of firm surrounding tissue in the tricuspid annulus, and the frequent annular dilatation in patients with tricuspid regurgitation. Percutaneous tricuspid valves also will not prevent further annular dilation and may therefore be susceptible to future dehiscence or embolization.
However, the greatest limitations facing tricuspid valve therapies are not the valve-related factors but, rather, the disease-related factors in this population, including right ventricular failure and advanced pulmonary hypertension.
Дата добавления: 2015-10-26; просмотров: 184 | Нарушение авторских прав
| <== предыдущая страница | | | следующая страница ==> |
| Endoscopic and minimally invasive surgery of heart defects | | | Відмінність фінансових інвестицій від реальних. |